- Home
- Forums
- National and International Business System Standards
- IATF 16949 - Automotive Quality Systems Standard
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
3 year audit cycle IATF 16949
- Thread starter xfngrs
- Start date
xfngrs
Quite Involved in Discussions
I used to have a table that showed which processes had to be audited once a year vs once per audit cycle. Does anyone know is this published somewhere? I don't find it in our latest 3rd party audit report.
I think this is determined by what you establish in your QMS Manual or system. It is based on risk to customer as well. Doing product audits annually is a given, customer score cards, and management responsibility to name a few. I hope that is helpful.
Thank you,
I used to have a table that showed which processes had to be audited once a year vs once per audit cycle. Does anyone know is this published somewhere? I don't find it in our latest 3rd party audit report.
Good day @xfngrs;
As already eluded to by others, there is no such requirement. Please clarify if you are speaking of...
QMS audits or...
Process audits or...
Product audits.
Regardless of which, there is no specified frequency (and certainly no specified "which process") requirement.
QMS and Process audits are required to cover "all" over a three year period. Both audit requirements include an "annual program", which simply means your organization must apply some auditing annually (can not wait until year three and then audit "all").
"Importance", "changes..." to the QMS/process, and results of "previous audit results" (i.e. risk) are the intended governance over audit frequency and priority.
Hope this helps.
Be well.
xfngrs
Quite Involved in Discussions
We are doing mostly QMS audits, but some audits of manufacturing lines as well.Good day @xfngrs;
As already eluded to by others, there is no such requirement. Please clarify if you are speaking of...
QMS audits or...
Process audits or...
Product audits.
Regardless of which, there is no specified frequency (and certainly no specified "which process") requirement.
QMS and Process audits are required to cover "all" over a three year period. Both audit requirements include an "annual program", which simply means your organization must apply some auditing annually (can not wait until year three and then audit "all").
"Importance", "changes..." to the QMS/process, and results of "previous audit results" (i.e. risk) are the intended governance over audit frequency and priority.
Hope this helps.
Be well.
As said for internal audits you have 3 years to audit all the system BUT
The audit programme shall be prioritized based upon risk internal and external performance trends, and criticality of the process(es).
The frequency of audits shall be reviewed and, where appropriate, adjusted based on occurrence of process changes, internal and external nonconformities, and for customer complaints. The effectiveness of the audit programme shall be reviewed as a part of management review.
The 3rd part audit needs to audit certain aspects every time as per the rules, I think that this is where you are confused
The audit programme shall be prioritized based upon risk internal and external performance trends, and criticality of the process(es).
The frequency of audits shall be reviewed and, where appropriate, adjusted based on occurrence of process changes, internal and external nonconformities, and for customer complaints. The effectiveness of the audit programme shall be reviewed as a part of management review.
The 3rd part audit needs to audit certain aspects every time as per the rules, I think that this is where you are confused
I used to have a table that showed which processes had to be audited once a year vs once per audit cycle. Does anyone know is this published somewhere? I don't find it in our latest 3rd party audit report.
Hi,
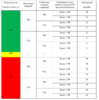
Here is a simple method for definition of audit frequency based on parameters required in clauses 9.2.1 and 9.2.2.1.
Enghabashy
Quite Involved in Discussions
I see that all system ,processes & products audits should covered yearly
*the frequency & amending the schedule should amend according to : the results of previous audits , the importance & assessment of process in Q.M system ; the recall / complaint received & any OEM /CSR requirements also ; including 2nd party audit results of OEM if applicable ,--etc.
*the 3 years cycle also are including that all records, schedules and documentation should be kept at least stated period also “ 3 years or more “ – this requirements is applicable to raise major NC if repeated during this cycle.
*the frequency & amending the schedule should amend according to : the results of previous audits , the importance & assessment of process in Q.M system ; the recall / complaint received & any OEM /CSR requirements also ; including 2nd party audit results of OEM if applicable ,--etc.
*the 3 years cycle also are including that all records, schedules and documentation should be kept at least stated period also “ 3 years or more “ – this requirements is applicable to raise major NC if repeated during this cycle.
Your three points contradict each other. Please see Howard's post above. The idea that all systems, processes and products should be audited yearly is a common misunderstanding among the standards.I see that all system ,processes & products audits should covered yearly
*the frequency & amending the schedule should amend according to : the results of previous audits , the importance & assessment of process in Q.M system ; the recall / complaint received & any OEM /CSR requirements also ; including 2nd party audit results of OEM if applicable ,--etc.
*the 3 years cycle also are including that all records, schedules and documentation should be kept at least stated period also “ 3 years or more “ – this requirements is applicable to raise major NC if repeated during this cycle.
I agree with your second point.
I do not fully understand your third point.
Similar threads
- Replies
- 11
- Views
- 2K
- Replies
- 6
- Views
- 363
- Replies
- 19
- Views
- 834
- Replies
- 14
- Views
- 1K
