plmiquelon
Registered
Hi,
I'm working on implementing a software verification and validation procedure for the small medical device company I work. We developed all PCB and software internally (1 person) for a Class I medical device that integrates a software to control some feature of the device. The software would be classified as class A based on IEC 62304 definition. I'm trying to follow the V-shape model
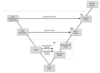
Here is what we have in mind:
1) Risk analysis for the whole medical device (product)
2) Requirement traceability matrix for the product that triggers software requirements
3) Risk analysis for the software
3) Requirement traceability matrix for the software
4) Produce a software architecture (input and output, logical structure (state machine)), and PCB architecture scheme (input-output, illustration of each PCB component and interactions). In these, we will identify the input and output and link the hardware and software input-output.
5) Verification : Confirmation that the requirements of the software have been addressed from a software and hardware standpoint (How would you do that?) - Integration and Implementation testing (IQ)
6) Validation : Multiple tests case that evaluates the behavior of the software-hardware depending on the input-output implemented (Expected results vs real results) - System testing (OQ)
7) After that, I would see a last step where we test the device itself with the software (Acceptance testing) but this remains difficult for me to define what we need to do exactly. The design verification-validation is clear to me from a hardware standpoint but new for software...
I'm looking for concrete steps, particularly from a software point of view. Reading multiple threads and website remains very vague.
Thank you!
I'm working on implementing a software verification and validation procedure for the small medical device company I work. We developed all PCB and software internally (1 person) for a Class I medical device that integrates a software to control some feature of the device. The software would be classified as class A based on IEC 62304 definition. I'm trying to follow the V-shape model

Here is what we have in mind:
1) Risk analysis for the whole medical device (product)
2) Requirement traceability matrix for the product that triggers software requirements
3) Risk analysis for the software
3) Requirement traceability matrix for the software
4) Produce a software architecture (input and output, logical structure (state machine)), and PCB architecture scheme (input-output, illustration of each PCB component and interactions). In these, we will identify the input and output and link the hardware and software input-output.
5) Verification : Confirmation that the requirements of the software have been addressed from a software and hardware standpoint (How would you do that?) - Integration and Implementation testing (IQ)
6) Validation : Multiple tests case that evaluates the behavior of the software-hardware depending on the input-output implemented (Expected results vs real results) - System testing (OQ)
7) After that, I would see a last step where we test the device itself with the software (Acceptance testing) but this remains difficult for me to define what we need to do exactly. The design verification-validation is clear to me from a hardware standpoint but new for software...
I'm looking for concrete steps, particularly from a software point of view. Reading multiple threads and website remains very vague.
Thank you!
